BRAND INFORMATION
Brand name: Wegovy
Active ingredients: Semaglutide
Schedule: S4
Manufacturer: Novo Nordisk
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using Wegovy
What is Wegovy?
Wegovy is a prescription weight-loss injection for individuals who are obese or overweight and have a medical condition directly connected to their excess weight.
For best results, use Wegovy with a calorie-restricted diet and regular exercise.
Due to the presence of semaglutide in Wegovy, it should not be used with any other GLP-1 receptor agonist medications.
It is unknown whether this drug interacts negatively or positively with other weight reduction supplements (prescribed, over-the-counter, or herbal).
It is also unknown whether Wegovy is safe for persons who have had pancreatitis in the past.
Wegovy’s effectiveness and safety in children under 18 have yet to be established.
Warnings and Precautions
Further evaluation is warranted if an elevated blood calcitonin level is found or if thyroid nodules are detected via a physical examination of the neck.
Serious and potentially deadly hemorrhagic or necrotising pancreatitis has been linked to semaglutide. Clinical trials of Wegovy® showed such results. Keep an eye out for signs of acute pancreatitis (including persistent severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting). Quit Wegovy® if acute pancreatitis is suspected.
1.6% of those on Wegovy® and 0.7% of those taking a placebo had cholelithiasis or gallstones. Cholecystitis affected 0.6% of those who took Wegovy® and 0.2% of those who took a placebo. People who used Wegovy® were more likely to have acute gallbladder sickness than those who took a placebo. It was true even after considering weight loss. Clinical evaluation and gallbladder testing are necessary in cases of suspected cholelithiasis.
Wegovy® may cause low blood sugar levels. Patients on Wegovy® were more likely to have hypoglycemia (6.2% vs 2.5%). Patients with type 2 diabetes on an insulin secretagogue (such as sulfonylurea) or insulin may be at a higher risk of hypoglycemia if they also take Wegovy®. Patients should know about the dangers of hypoglycemia and what to do if they experience it. Those with type 2 diabetes should keep an eye on their sugar levels.
People who have taken semaglutide have reported experiencing acute kidney injury and increasingly severe chronic renal failure, leading to the need for hemodialysis. Acute kidney injury has occurred in some patients without preexisting renal disease. Patients who had been experiencing severe nausea, vomiting, or diarrhea and had lost a significant amount of fluids were disproportionately affected.
Patients who report severe gastrointestinal reactions to Wegovy® or renal impairment or those who experience adverse events that might lead to fluid depletion should have their renal function monitored throughout initiation or increase in dose.
Extremely sensitive reactions to Semaglutide have been reported (anaphylaxis, angioedema). If hypersensitivity responses occur while taking Wegovy®, quit the drug immediately and seek medical attention.
GLP-1 receptor agonists should be used cautiously in individuals with anaphylaxis or angioedema. Retinopathy caused by diabetes complications from diabetes: researchers found that 4.0% of those given Wegovy® experienced diabetic retinopathy, compared to 2.7% of those given a placebo. Short-term improvements in diabetic retinopathy may be achieved with aggressive glucose management. Patients with diabetic retinopathy should be monitored for any signs of worsening.
Increasing heart rate. Wegovy® participants in clinical trials reported a 1 to 4 bpm greater resting heart rate than placebo patients. Patients on Wegovy® had a higher prevalence of 10–19 bpm and 20 bpm+ baseline changes compared to placebo. Contact your doctor immediately if you have palpitations or a fast heartbeat at rest. When patients’ resting heart rates increase, they stop Wegovy® treatment.
Clinical trials of many weight-loss medications have shown increased suicidal ideation and behaviour. Take note of changes in mood that could indicate depression. Discontinue Wegovy® in those who have attempted suicide or who are currently suicidal.
Who should not use Wegovy?
You should not use Wegovy if:
- you or any of your family have ever had a type of thyroid cancer called medullary thyroid carcinoma (MTC), or if you have an endocrine system condition called Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- you have had a serious allergic reaction to semaglutide or any of the ingredients. See the end of this page for a complete list of ingredients. Symptoms of a serious allergic reaction include:
- swelling of your face, lips, tongue or throat
- problems breathing or swallowing
- severe rash or itching
- fainting or feeling dizzy
- very rapid heartbeat
Wegovy pregnancy and breastfeeding warnings (more details)
Drug Interactions
- Wegovy® hasn’t been tested in insulin-treated individuals. Lessen insulin secretagogues (such as sulfonylureas) or insulin doses while starting Wegovy® to reduce hypoglycemia risk.
- Wegovy® delays stomach emptying and may affect oral drug absorption. Wegovy® may interact with oral medicines.
- Wegovy slows digestion and absorption of oral medications.
- Tell your doctor about all your medications, including insulin, dulaglutide, exenatide, liraglutide, Byetta, Trulicity, Victoza, and others.
- Other medications, vitamins, and herbal items may interact with Wegovy. Let your doctor know about all your medications.
- Wegovy drug interactions (more details)
How should I use Wegovy?
Read the Instructions for use that come with your prescription.
- Use Wegovy exactly as your healthcare provider tells you to.
- Your healthcare provider should show you how to use Wegovy before you use it for the first time.
- Wegovy is injected under the skin (subcutaneously) of your stomach (abdomen), thigh, or upper arm. Do not inject Wegovy into a muscle (intramuscularly) or vein (intravenously).
- You can take Wegovy with or without food.
- Taking too much Wegovy, you may have severe nausea, vomiting, and low blood sugar. Call your healthcare provider or go to the nearest emergency room immediately if you experience any of these symptoms.
Learn the detailed Wegovy dosage information before you start using this medication.
You should immediately:
phone the Poisons Information Centre
(by calling 13 11 26 in Australia or 0800 764 766 [0800 POISON] in New Zealand), or
contact your doctor, or
go to the Emergency Department at your nearest hospital.
Wegovy side effects
Wegovy may have severe adverse effects.
Pancreatitis. Stop taking Wegovy and notify your healthcare practitioner if you experience serious stomach discomfort. The ache may be in your abdomen and back.
Gallbladder issues. Wegovy may induce gallstones. Some gallbladder issues are surgical. Call your doctor if you develop these symptoms:
- tummy ache (abdomen)
- skin or eye yellowing (jaundice)
- clay stool
Increased risk of low blood sugar (hypoglycemia) among type 2 diabetes patients, particularly those using sulfonylureas or insulin. Low blood sugar in type 2 diabetics using Wegovy may be dangerous. Consult your doctor about low blood sugar. Check it before and throughout therapy. Signs of low blood sugar include:
- wooziness
- haze
- irritation, anxiety, or mood changes
- slouched speech
- hunger
- feeling confused or tired
- shakiness / weakness
- headache
- fast heartbeat
Kidney issues (kidney failure). In persons with renal difficulties, diarrhea, nausea, and vomiting may induce dehydration, worsening their condition. Consume fluids to avoid dehydration.
Stop taking Wegovy and obtain medical care if you develop an allergic response, including swelling of your face, lips, tongue, or throat, breathing or swallowing, rash or itching, fainting or dizziness.
Vision changes with a fast pulse in type 2 diabetes. Changes in eyesight during therapy should be reported.
High heart rate. Wegovy raises heart rate during rest. Your heart rate should be monitored when using Wegovy. Tell your doctor if your heart races or pounds for many minutes.
Suicidal depression. Mind changes, particularly unexpected mood, behaviour, cognition, or emotion changes should be monitored. Call your doctor if you develop new, severe, or worrying mental changes.
Common side effects:
- nausea
- diarrhea / vomiting
- constipation
- stomach (abdomen) pain
- headache
- weariness (fatigue)
- nausea dizziness
- bloated gassy
- flu heartburn
Discuss any bothersome or persistent side effects with your doctor. There are other adverse effects.
Call your doctor for adverse effects. TGA’s hotline is 1300 134 237.
More details on Wegovy side effects are here.
What are the ingredients in Wegovy?
Active Ingredient: semaglutide
Inactive Ingredients: disodium phosphate dihydrate, sodium chloride, and water for injection
Appearance
Instructions for Use
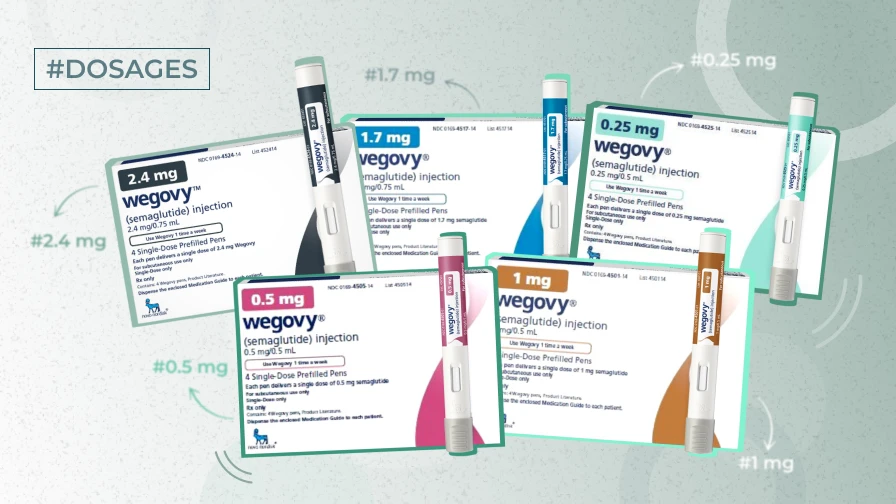
There are five Wegovy strengths. Talk to your healthcare physician or caregiver before using Wegovy for the first time.
Read Wegovy’s instructions first. This info isn’t a substitute for talking to your doctor.
- Wegovy pens are single-use. Wegovy is used subcutaneously.
- Your pen contains a Wegovy dose.
- The needle cover hides it.
- Before injecting, remove the pen cap.
- Don't touch the needle cover. Needlestick injuries are possible.
- When the needle cover touches your skin, the injection begins.
- Do not remove the pen before the yellow bar stops moving. You may not receive the complete dosage if the needle is withdrawn early.
- When withdrawn from the skin, the needle cover locks. You can't pause and resume injection.
- Blind or visually impaired people shouldn't utilise the Wegovy pen without help.
If the yellow bar doesn’t move or stops during injection, call your doctor or Novo Nordisk at startWegovy.com or 1-833-934-6891.
Wegovy pen instructions
Don’t use your pen without medical training. Before starting therapy, be sure you or your carer can use the pen. Read and follow the pen instructions.
Step 1. Prep your injection Wegovy injection supplies:
- 1 alcohol swab or soap and water
- 1 gauze pad or cotton ball
- 1 disposable container
- Wash your hands.
- Check your Wegovy pen.
Do not use your Wegovy pen if:
- The pen seems worn or damaged as if it dropped.
- Wegovy medication isn’t clear and colourless through the pen window.
- EXP has passed.
If your Wegovy pen fails these tests, call Novo Nordisk at 1-833-934-6891.
Step 2. Choose an injection site
- Your doctor may recommend an injection location.
- Upper legs (front thighs) or lower stomach (keep 2 inches away from your belly button).
- Someone else can provide the upper-arm injection.
- Never inject into fragile, bruised, red, or firm skin. Avoid scars and stretch marks.
- Each week, inject in the same body region but not in the same location.
- Use alcohol or soap to clean the injection site. After cleaning, don’t touch the injection site.
Step 3. Cap pen
- Remove your pen’s cap.
Step 4. Inject Wegovy
- Push the pen until the yellow bar stops moving.
- Press the pen harder if the yellow bar doesn’t move.
- You will hear 2 clicks during the injection.
- Click 1: injection begins.
- Click 2: injecting.
Step 5. Discard pen
After usage, discard the Wegovy pen.
How can I discard Wegovy pens?
Blood after the injection?
Using gauze or cotton, softly press the injection site if blood occurs.
How can I discard Wegovy pens?
After usage, put the Wegovy pen in a sharps container. Don’t trash the pen.
You may use a home container that is:
- plastic, heavy-duty;
- sharps-proof, with a tight-fitting, puncture-resistant cover;
- standing firm;
- leak-proof;
- appropriately identified as hazardous garbage.
When your sharps container is nearly full, follow your community’s disposal standards. State or municipal legislation may govern how to dispose of needles and syringes. Please check out the additional information about safe sharps disposal.
- Do not reuse the pen.
- Do not recycle the pen or sharps container.
Keep your Wegovy pen, sharps container, and medications away from kids.
What is the best pen care?
Pen safety:
- Don’t drop or hit your pen.
- Avoid liquids near your pen.
- Do not mend a broken pen. Use a new one.
- Before injecting, replace the pen cap. If you keep an unused pen without the cap, remove and replace it, or lose it, the pen is no longer sterile. This might result in infection.
Who distributes
Novo Nordisk is the only firm that has clearance from the TGA to commercialise Wegovy®. They deliver it in a pen that is disposable after a single use and is only accessible by prescription.
References
- What is the most important information I should know about Wegovy? Available from: [URL_Link]
- WEGOVY (Novo Nordisk Pharmaceuticals Pty Ltd). Available from: [URL_Link]
- NOVOMEDLINK. Obesity. Important Safety Information for Wegovy® (semaglutide) injection 2.4 mg. Available from: [URL_Link]
- Updates about Wegovy®. Available from: [URL_Link]
- Wegovy (semaglutide): a new weight loss drug for chronic weight management. Available from: [URL_Link]
- What is Wegovy? Available from: [URL_Link]
- What is Wegovy? Important information. Available from: [URL_Link]
Common questions about Wegovy
No, the company that makes Wegovy hasn’t shared reviews from people who’ve used it.
You can find out more about what to expect from Wegovy by going to the website. Or you can read the drug’s prescribing information.
For more information, talk with your doctor or pharmacist.
Wegovy, Ozempic, Trulicity, and Victoza are all glucagon-like peptide-1 (GLP-1) agonists, one group of drugs. But they are not the same in several ways.
The active substance semaglutide is in both Wegovy and Ozempic. The active ingredient of Trulicity is called dulaglutide, and the active drug in Victoza is liraglutide.
The TGA has approved Ozempic, Trulicity, and Victoza to treat type 2 diabetes.
This is not a good way to use Wegovy. Instead, Wegovy is approved for long-term weight control in certain adults when used with workouts and a low-calorie diet. (See “Wegovy for weight loss and weight management” above for more information.)
Talk to your doctor or pharmacist to find out how Wegovy differs from Ozempic, Trulicity, and Victoza.
The company that makes Wegovy has not shown before-and-after pictures of people who have taken the drug. Talk to your doctor about what to expect from Wegovy to learn more.
Wegovy is not used to treat either type 1 or type 2 diabetes. Wegovy is recommended for long-term weight control in some individuals. (For further information, check the section above under “Wegovy for weight reduction and weight control.”)
However, semaglutide (the active ingredient in Wegovy) is also available under the trade names Ozempic and Rybelsus. Ozempic and Rybelsus are both medications used to treat type 2 diabetes. There is presently no approved version of semaglutide for treating type 1 diabetes.
Consult your doctor if you want to use different kinds of semaglutide to treat your diabetes.
It’s unlikely. It is unknown if Wegovy is safe or effective when used with other weight reduction and control medications. Consult your doctor before using Wegovy with any drugs, vitamins, herbs, or weight-loss products.
Wegovy should be taken in conjunction with a diet reduced in calories and frequent exercise. Some folks may also benefit from behavioural therapy or dietician advice. Along with Wegovy, see your doctor about alternative safe methods to control your weight.
Other medications for weight reduction and control are available. Some may be more suitable for you. If you’re looking for an alternative to Wegovy, consult with your doctor. They may advise you on different drugs that may be beneficial to you.
Examples of other drugs that may be used for weight loss and weight management include:
- benzphetamine
- bupropion and naltrexone (Contrave)
- liraglutide (Saxenda)
- orlistat (Alli, Xenical)
- phendimetrazine (Bontril PDM)
- phentermine (Adipex-P, Lomaira)
- phentermine and topiramate (Qsymia)
Disclaimer
These drug details aren’t for emergencies. If you’re unwell, have an overdose, or face an emergency, call triple zero (000).
It was created with accuracy and attention to detail. This content should not be used in lieu of medical advice, diagnosis, or treatment to manage or diagnose a medical issue. Our terms and conditions include further info.












One of the best weight-loss drugs I’ve tried!
Hi, I was just wondering how you were able to purchase Wegovy, I thought there was no stock, any chance of helping me out, or guiding me in the right direction?
The thing about consulting a doctor is the main point. No matter how much theory you know, it doesn’t make you a pro!
This information was quite useful for me. I am just about to begin taking the medication. Thanks!
Oh… side effects sound so scary. I hope I will be lucky not to have them.
I read the pen instructions 10 times! Was so afraid, but I did my injection for the first time. Thanks for the detailed explanations.
Good job! Before using this, absolutely everyone should be aware of this!!
I’m curious to learn more about how this medication and eyesight alterations are related. Why are diet medication and eye illnesses connected?